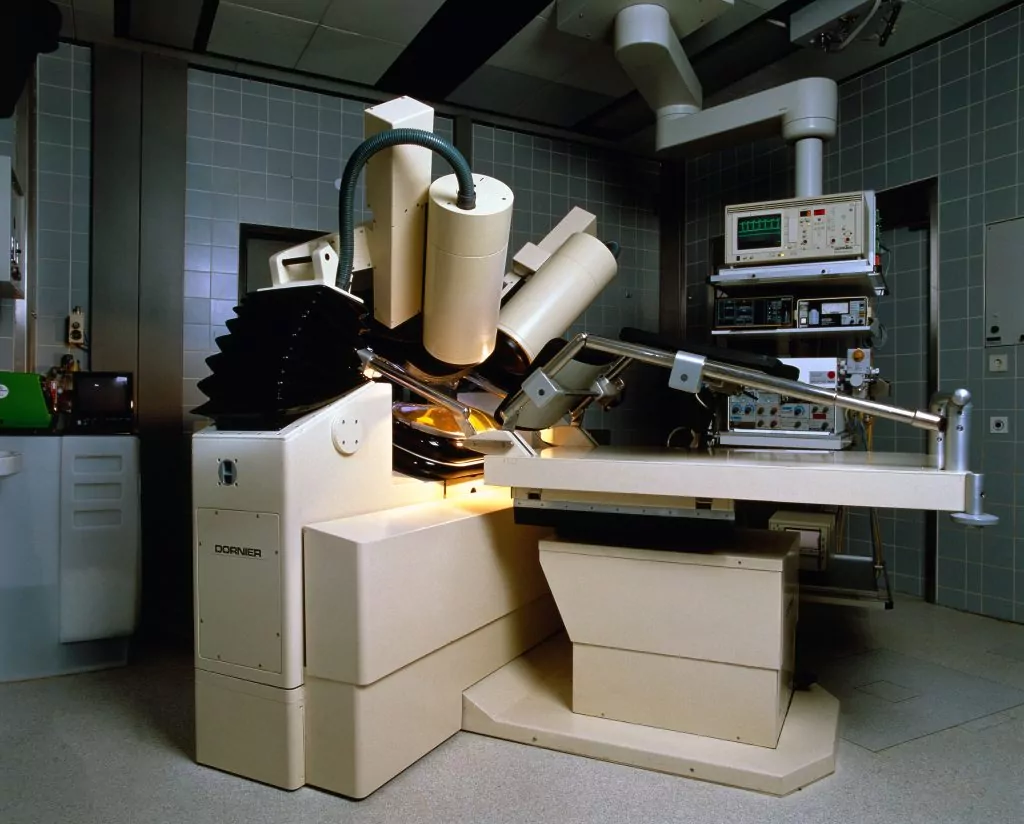
An Important Day in Medical History
On February 7, 1980, doctors performed the first ESWL lithotripsy treatment on a patient with kidney stones. Many procedures later, the treatment proved so successful, that it revolutionized modern kidney stone management. Based on extensive research pioneered by Dornier MedTech, lithotripsy is now used on 500,000+ kidney stone patients each year.
Based on decades of experience, the treatment is popular due to its non-invasiveness and reliability, and that it reduces the recovery time compared to surgical options. If you have kidney stones, or know someone who does, learn more by clicking here to visit our Patient Center!
About Dornier MedTech – Dornier MedTech, a wholly-owned subsidiary of Accuron Medical Technology group (www.accuron.com), is a medical device company focused on providing leading technology and improving life by delivering scientifically superior products and solutions to physicians and patients involved in urological care. As pioneers of the lithotripsy and a variety of surgical lasers, Dornier’s 40 years of innovation and service has made it one of the most trusted MedTech companies in the industry. Dornier MedTech is headquartered in Munich, Germany with offices and distributors all over the world. For more information, visit www.dornier.com.
About the Accuron Medical Technology Group – The Accuron Medical Technology Group is a privately owned by Temasek Holdings, through the business of Dornier MedTech GmbH, Veredus Laboratories and Advanced Materials Technologies Pte Ltd, provide therapeutic, diagnostic, and medical device manufacturing solutions to customers around the globe. Veredus Laboratories Pte Ltd, specializes in the development, manufacture, and marketing of innovative molecular diagnostic solutions in the clinical, specialty, and custom testing markets based on a proprietary Lab-on-Chip platform. Advanced Materials Technologies Pte Ltd serves the contract manufacturing needs of the medical technology sector through providing materials expertise and high precision engineering capabilities.